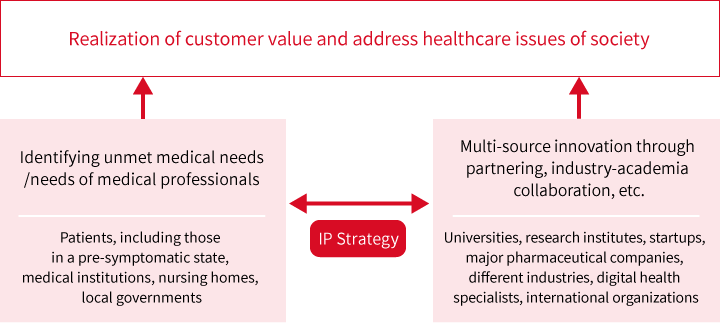
IP Strategy
SHIONOGI Group’s Philosophy and Intellectual Property/Intangible Asset Strategy
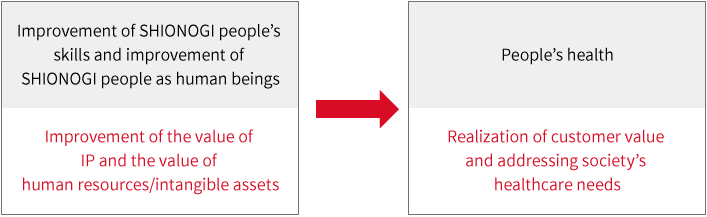
SHIONOGI Group is committed to its Heritage (The Company Policy of Shionogi) to “strive constantly to supply the best possible medicine to protect the health and wellbeing of the patients we serve.” As the goal of its corporate activities, and for this purpose, SHIONOGI people will need to “strive ceaselessly day after day to improve their skills” and “strive ceaselessly day after day to improve as human beings.”
SHIONOGI Group believes that improving the value of its technology and intellectual property, as well as the value of its human resources and intangible assets, will lead to realizing customer value and to addressing the healthcare needs of society.
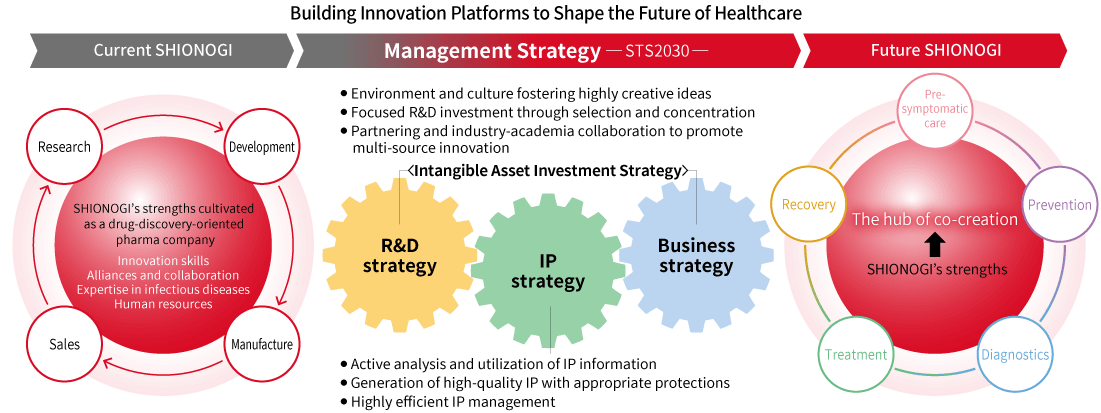
SHIONOGI Group’s Management Strategy and Intellectual Property/Intangible Asset Strategy
The SHIONOGI Group, in order to realize its 2030 Vision (Building Innovation Platforms to Shape the Future of Healthcare), carefully aligns its intangible asset investment strategy and intellectual property strategy as the two core elements supporting the Management Strategy of SHIONOGI Group as it evolves toward becoming a Healthcare as a Service (“HaaS”) company.
Intellectual property and intangible assets are not limited to intellectual property rights, such as patent rights, trademark rights, design rights, and copyrights. They include a wide range of assets, such as technologies, brands, design, content, data, know-how, customer networks, trust/reputation, value chains, and supply chains, as well as the organizational capabilities and processes to create them.
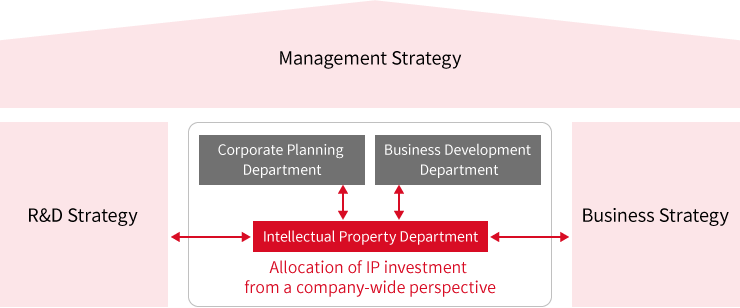
Implementation of Intangible Asset Investment Strategy and Intellectual Property Strategy
1.Intellectual property governance system and R&D investment
SHIONOGI Group closely aligns its R&D, business, and intellectual property strategies so that priorities and focus areas are clearly synchronized.
From the perspective of a company-wide investment in global intellectual property assets, we have established the Patent Management Guidelines and the Trademark Management Guidelines to define our policies for investment in patents and trademarks related to each R&D or business investment program. According to these guidelines, application preparation, maintenance and management are aligned with the responsible personnel from each global value chain.
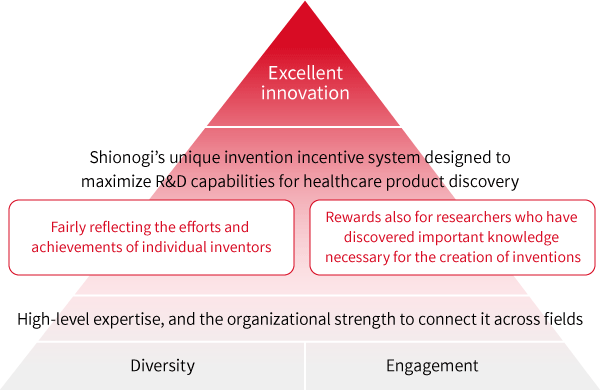
2.Fostering a culture to promote R&D activities
Based on the belief that diversity and engagement will contribute significantly to the number and value of inventions generated from our research, we have established an invention incentive system unique to Shionogi.
1) Shionogi’s high expertise across a range of scientific fields, and the organizational strength to connect them, greatly contribute to our ability to create new healthcare solutions. In light of this, our incentive system is designed to reward not only inventors but also those who have discovered important knowledge necessary for the creation of inventions in a well-balanced manner.
2) By providing rewards to inventors who have contributed to progress in the early stage of product development, the system is designed to fairly allocate these rewards across research and therapeutic areas, isolating them from the impact of factors irrelevant to their efforts or achievements, such as cancellation of development due to strategic reasons or commercial factors such as the market environment at the time of launch.
In this way, Shionogi’s unique invention incentive system is designed to maximally incentive our R&D capabilities for healthcare product discovery and help foster the ground for innovation.
3.Promoting multi-source innovation through partnering with industry and academia
At present, the SHIONOGI Group has established partnerships with over 50 organizations as described in “Partnering” of the “Innovation” section of our website. (See Partnering)
Collaborations can start in the earliest stages of exploratory research, or at later stages, to support innovation.
With a view to initiating new businesses, for example in the HaaS and digital health fields, and to identify and pursue partnerships with academia and companies across different industries, as well as to identify individual and societal healthcare needs and changes therein, we use sophisticated IP landscape analysis techniques.
* IP landscape analysis: the process of obtaining information useful for formulating R&D and business strategies based on integrated analysis of both intellectual property and non-intellectual property information
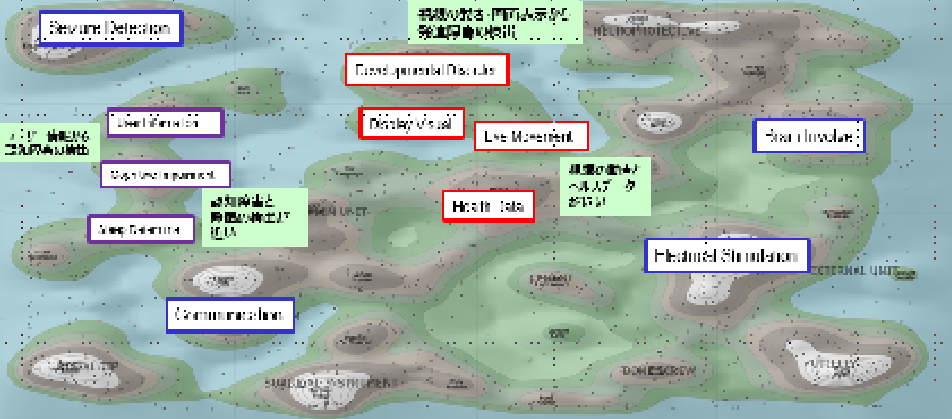
4.Active analysis and utilization of intellectual property information
At SHIONOGI Group, we use IP landscape analysis to gain an overall view of the R&D areas, develop innovation strategies, search for alliance partners, identify opportunities for drug repositioning, and other similar activities, thereby contributing to the formulation and deployment of R&D and business strategies.
The goal of IP landscape analysis at Shionogi is:
to provide overall information regarding fields in which we are present or are considering entering to formulate and strengthen our R&D and business action plans.
To achieve this, Shionogi focuses on the three elements below:
1) Linking IP/technology information to healthcare needs
2) Predicting the evolution of research fields and markets
3) Improving our strategic decision-making and actions

Regarding Shionogi’s IP landscape analysis initiatives, at an opinion exchange meeting with the Commissioner of the Japan Patent Office (JPO), our President confirmed the principle that “Intellectual property is the basis of management of a pharmaceutical company.” The Intellectual Property Department at Shionogi plays the internal consulting role, aligned with the Management Strategy, to achieve our overall goals.
In the “Report for the Study of the Analysis and Utilization of Intellectual Property Information Contributing to Management Strategies” (pp 302-304) of JPO, Shionogi’s IP landscape analysis initiative was introduced. (See chizai-jobobunseki-report.pdf (jpo.go.jp))
The image on the left shows a landscape map of technologies related to digital diagnosis in the CNS area (edited and cited from Derwent Innovation), which was utilized as a tool to analyze recent R&D trends in this field.
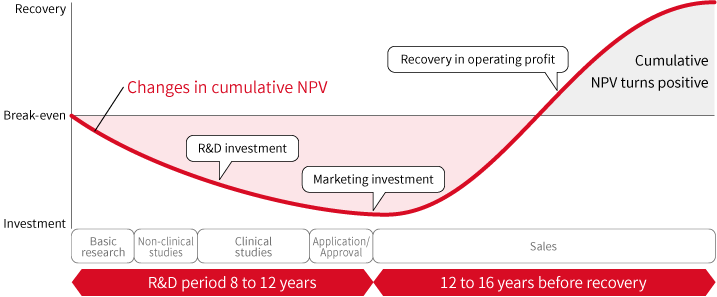
5.Basic patent strategy
The R&D of a new drug is particularly challenging activity, facing many hurdles such as low probability of success, protracted R&D period, and enormous R&D costs. In such an environment, in order to grow as a company while contributing to society by continuously offering innovative new drugs, it is crucial to recover the R&D investment made and to be able to invest in further R&D. To this end, SHIONOGI Group believes that it is necessary to properly and appropriately protect our innovation with IP. (See Shionogi Group Intellectual Property Policy.)
6.Securing appropriate patent rights
Unlike in the electrical and mechanical fields, which are technological fields where products are often protected with hundreds to thousands of patents, products in the pharmaceutical and biological fields are often protected by fewer patents. Therefore, the value of each patent in these fields may be greatly different from that in other fields.
Therefore, the SHIONOGI Group, pursues high-quality patents with a scope of protection properly reflecting our innovation, while working to extend the protection period, where appropriate, utilizing the patent period extension system for pharmaceutical patents in various countries around the world.
Moreover, innovations generated in subsequent R&D processes (uses, crystal forms, manufacturing methods, formulation patents, etc.) are also protected properly as intellectual property.
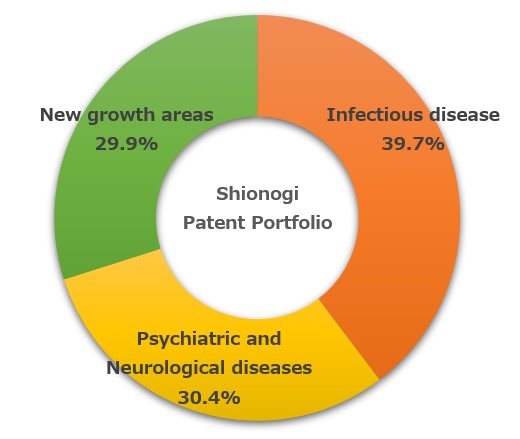
As a result of these efforts to obtain IP rights, the percentages of the number of patents (patent applications) we have obtained in our R&D focus areas of infectious disease, psychiatric and neurological diseases, and new growth areas are shown in the pie chart on the right.
Global applications account for 90.7% of all applications*1 and the percentage of inventions applied for overseas*2 has reached as high as 77.8%.
* Both figures are as of the end of March 2024 and applications filed only in the name of partners are not included.
*1 : (1 - The number of Japanese patents / The number of worldwide patents) x 100(%)
*2 : (The number of PCT filed patent applications based on the number of first priority patent applications in 2022 / The number of first priority patent applications in 2022) x 100(%)
7.Maintenance of patent portfolio, prevention of infringement, and exercise of rights
While reviewing the patent portfolio as necessary according to our business strategy, we periodically monitor for potential infringement and consider the appropriate way to exercise our rights, including through licensing agreements and, if necessary, legal action. Through these measures, we seek to make effective use of our full IP portfolio. We also respect valid intellectual property of third parties.
The left graph shows our yearly IP-related royalty income.
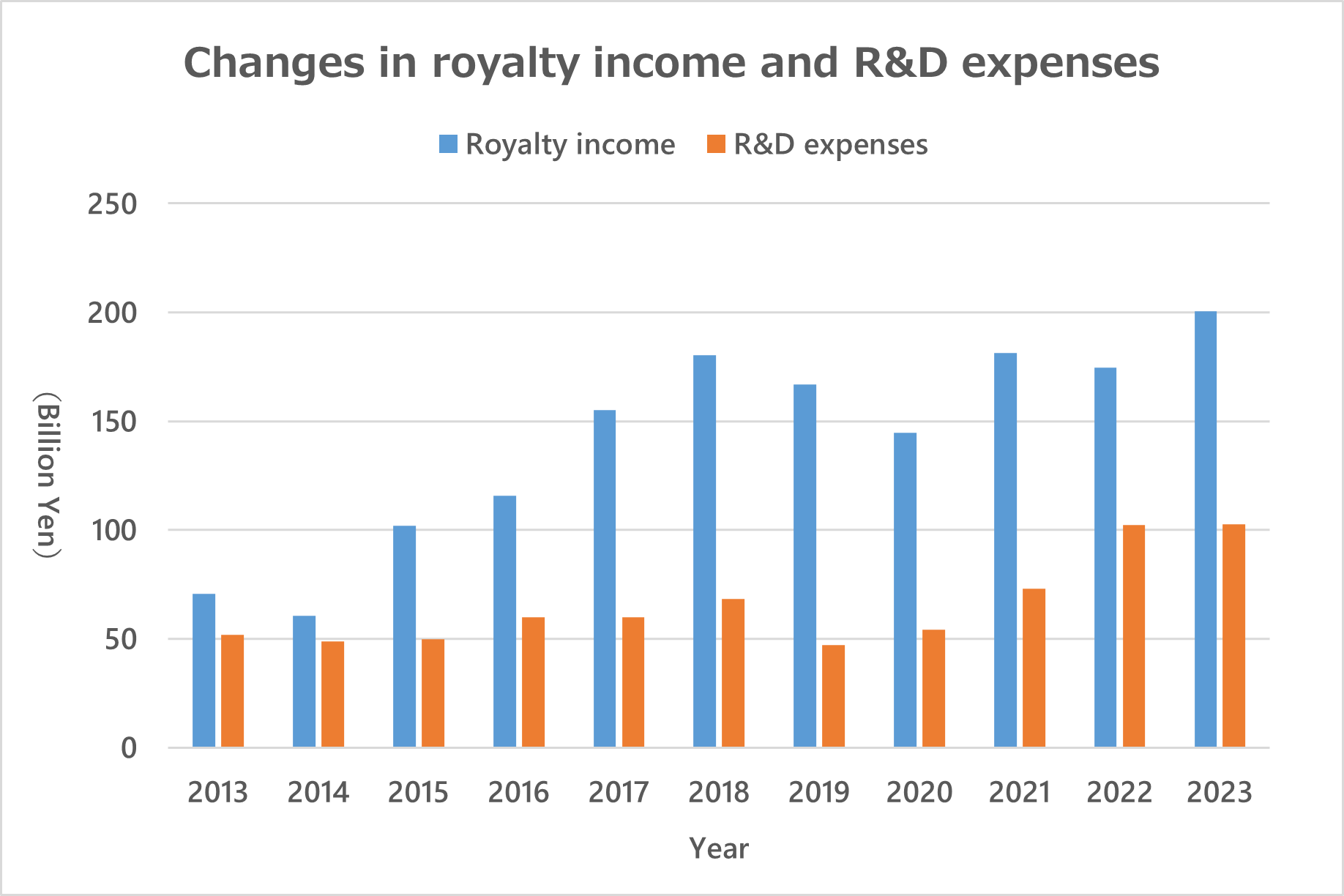
8.Brand & design strategy

SHIONOGI Group seeks to deliver pharmaceutical products to patients while establishing relationships of trust with physicians and medical institutions, as well as customers and society more broadly. Meanwhile, in accordance with our vision to transform ourselves to a HaaS company in accordance with STS2030, we aim to provide total healthcare solutions, not only addressing health problems when they arise, but seeking to prevent them from arising in the first place.
As the healthcare business evolves more and more toward a comprehensive service offering rather than individual products, the brand & design strategy becomes more important than ever. We therefore pursue appropriate trademark management based on the principles below.
1) Improvement of awareness and recall in a digital environment
2) Development of products/services taking into consideration UX (user experience) and UI (user interface)
3) Consideration for the diverse values of a wide range of global stakeholders
As part of our branding activities, we have established Shionogi Group’s Brand Trademark Management Rules and pursue our efforts on trademark management corresponding to our branding and globalization approaches.
9.Intellectual property education
Since the 2000s, SHIONOGI Group has engaged in intellectual property education efforts throughout our company, including in the R&D and management divisions, in order to effectively and correctly implement our global intellectual property strategy in close alignment with our R&D and business strategies.
These efforts focus on practical education encompassing numerous examples drawn from our industry and covering all the stages of drug R&D, rather than just explaining general principles based on patent laws. We plan and implement educational programs accommodating both a wide range of technical fields (such as chemical compounds, manufacturing methods, uses, crystals, and formulation), and employee experience levels (ranging from new entrants, employees in the second or third year after joining the company, and mid-level employees).
We also offer specialized courses to educate employees about the IP systems and requirements in our global markets outside Japan.
In view of the recent advancements in digital health, we have started new intellectual property education programs for digital health-related activities, while also enhancing courses on design, trademarks, copyrights (content), and trade secrets (including data and know-how) to reflect our increasing activities in the digital health area.

10.Improving access to pharmaceuticals
Since infectious disease, which has been a longtime focus of Shionogi, represents a global scourge creating innumerable losses of lives and livelihood, we understand the importance of ensuring that patients and medical professionals around the world have access to the drugs they need for the treatment of these diseases. However, the barriers to such access are not solely economic or IP-related. Supply chain, infrastructure, education, and other factors also have major impacts on access.
With respect to IP itself, our principles are outlined in our Shionogi Group Intellectual Property Policy (see Shionogi Group Intellectual Property Policy), including the policies below:
1) Limiting target countries/regions for patent applications or exercise of rights
2) Providing licenses using patent pools, and similar models
3) Participating in the Patent Information Initiative for Medicines (Pat-INFORMED)
Furthermore, as presented in the Shionogi Group Global Health Access Policy (See Shionogi Group Global Health Access Policy), we are working to establish an environment in which the SHIONOGI Group’s drugs are accessible for patients in need around the world.



